One of the most prominent features of plant cells is their cell wall. While many organisms produce some kind of extracellular matrix that is important in cell function, almost every cell made by a plant is surrounded by a rigid mixture of cellulose and other polysaccharides and proteins. The cell wall allows the formation of high hydrostatic pressure known as turgor pressure.
Cellulose
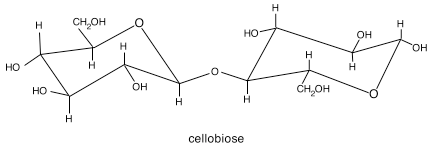
Cellulose is the major load-bearing structural component of the cell wall. In chemical terms, cellulose is β1→4-linked glucose dimers called cellobiose. These dimers repeat in long chains, which are crystallized with other such chains during synthesis to form microfibrils. Think of a microfibril as yarn, and individual chains as the threads that are spun into a yarn.
One of the peculiarities of cellulose in the cell wall is that it is not synthesized inside the cell and deposited in the apoplast by vesicles. Rather, cellulose is synthesized by a complex spanning the plasma membrane and extruded into the apoplast. These rosette-shaped structures, called terminal complexes, are made up of many individual cellulose synthase enzymes working in concert to produce a microfibril. Terminal complexes travel along a network of cortical microtubules that determine the pattern of deposition of cellulose microfibrils. Direct evidence for this hypothesis came from visualizing the interaction between microtubules and a part of the cellulose synthase enzyme complex.

Other Wall Components
In addition to cellulose, plant cell walls include a wide variety of polysaccharides and structural proteins. The polysaccharides include hemicelluloses and pectins, both of which are diverse groups of complex, branched carbohydrates. Unlike cellulose, neither of these constituents is manufactured directly in the cell wall, but both follow the more traditional intracellular synthesis followed by secretion. These polysaccharides function to stabilize the interaction between cellulose microfibrils through H-bonding.

Pardez AR, Somerville CR, Ehrhardt DW (2006) Visualization of Cellulose Synthase Demonstrates Functional Association with Microtubules. Science 312:1491-1495 See a collection of time-lapse movies.


