Category Archives: Germination
Germination
How do plants regulate germination?
- Seed Structure: What are the parts of a seed, and how are they organized to protect and nourish the embryonic plant?
- Dormancy & Germination: What is dormancy, and how is it regulated?
- Hormone perception: What role do hormones play in regulating dormancy?
- Phytochrome: What role does light play in regulating germination?
Phytochrome
Phytochrome
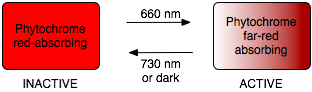
Phytochrome is known as a photoreversible photoreceptor, meaning that it can change forms from the inactive to active states (and vice-versa) by absorbing distinct wavelengths of light. When it absorbs red light, the phytochrome complex changes shape and becomes activated. At the same time, this change in shape means that it no longer absorbs in the red part of the spectrum, but its absorbance maximum has shifted to the far-red part of the spectrum. Interception of a photon in the far-red energy range will cause the complex to change shape back to the red-absorbing form, as will a prolonged period in the dark.
When embryo-dormant seeds with thin seed coats are exposed to red light in the environment, they are induced to begin germinating through the activation of phytochrome. And because phytochrome is photo-reversible, germination can be halted if the embryo detects that it is in the presence of lower energy far-red light. This watershed observation about germination was one of the first indications of the existence of the phytochrome pathway during the 1950’s and 1960’s:
Seed hit with red light germinated unless it was then hit with far-red; but if red again ensued, it would germinate. Incredibly, all that mattered was which color came last even if the seed was struck by 100 alternating cycles of red and far-red.
Phytochrome structure
Functional phytochrome molecules are composed of a protein component with a pigment molecule bound tightly to it. The apoprotein, as it is known, is encoded by one of five genes in Arabidopsis, and the chromophore, as the pigment is known, is a bilin-type pigment similar in structure to a chlorophyll precursor. Furthermore, phytochromes pair up with another phytochrome to form a dimer. This interaction happens between amino acids at the amino-terminal end of the protein. The other end of the protein, the carboxy-terminal end, shows homology to bacterial histidine kinase domains, but it isn’t clear whether phytochromes act as kinases themselves when activated.
Phytochrome signaling
Upon perception of red light, phytochrome molecules undergo a conformational change and appear to traffic to the nucleus, where they can interact with partner proteins. One such partner is PIF3 (Phytochrome Interacting Factor 3), a basic helix-loop-helix transcription factor that binds to the promoter of light-regulated genes. At least several of the genes regulated by PIF3 encode transcription factors themselves. In addition, phytochrome has been shown to increase expression of at least one gene encoding an enzyme in the GA biosynthetic pathway. GA biosynthesis, in turn, can promote the expression of hydrolytic enzymes associated with energy mobilization and uptake by the embryo.
Reference
Toyomasu et al. (1998) Phytochrome Regulates Gibberellin Biosynthesis during Germination of Photoblastic Lettuce Seeds. Plant Physiol 118: 1517-1523
Hormone Perception
Introduction
As previously discussed, high levels of ABA promote embryo dormancy, while a reduction in ABA concentration leads to an increase in GA biosynthesis and the promotion of germination. But how does the concentration of small molecules make a difference in the cell? These molecules have specific cellular receptors, capable of tightly binding the hormone and activating a signal transduction cascade. The end results of the cascade differ depending on the hormone, but almost always lead to a change in gene expression.
ABA perception
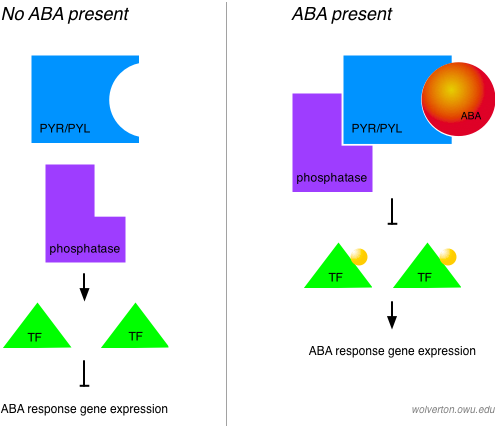
At least one ABA receptor has been identified to date in plants. The PYR1 gene encodes a protein that can bind ABA and inactivate a protein phosphatase in the PP2C class. Protein phosphatases remove phosphate groups from target proteins, often inactivating them. The result of inactivating this phosphatase is to stimulate ABA responses, the earliest of which include activation of ABA-specific transcription factors. So we can think of ABA as inhibiting an inhibitor of ABA signaling — when the inhibitor (a PP2C protein) is inhibited, ABA signaling can proceed.
A receptor for ABA has been sought for many years, but had eluded researchers until recently. The PYR1 gene was isolated in a screen for mutants that showed resistance to a synthetic compound called pyrabactin, an agonist of ABA responses. This kind of approach, called chemical genetics, is one way to side-step the functional redundancy shown by many plant genes due to genome duplication, and is described in more detail by the Cutler Lab, which successfully employed this approach to uncover PYR1.
GA perception
Recent evidence suggests that a protein called GID1 acts as a GA receptor. When GA binds to GID1, it causes the targeted degradation of a group of proteins known as the DELLA family that normally represses GA responses. By getting DELLA proteins out of the way, the cell can begin to mount a response to the presence of GA. This system of targeted degradation uses a pathway known as the 26S proteasome, a large, multi-subunit cellular garbage disposal. To guard against accidental recycling of proteins, the garbage disposal only accepts proteins with a special tag attached. The tag is itself a small protein called ubiquitin, and the closely regulated process of adding the tag to a target protein is called ubiquitination. The GA receptor GID1 is itself a part of the ubiquitinating machinery and acts by causing the ubiquitination of DELLA proteins. To help visualize this process, I have created an animation showing the interaction of these participants in GA signaling.
Responses to these 2 hormones during dormancy and germination are representative of 2 kinds of signal transduction pathways — kinase cascades and ubiquitin-mediated proteolysis. Both kinds of pathways play crucial roles across the domains of life in mediating responses to signals, and there are many more examples of each within the plant kingdom.
References
-
S. Y. Park, P. Fung, N. Nishimura, D. R. Jensen, H. Fujii et al. (2009) Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 324, 1068-1071.
-
Y. Ma, I. Szostkiewicz, A. Korte, D. Moes, Y. Yang et al. (2009) Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 324, 1064-1068.
-
Ueguchi-Tanaka, M. et al. (2005) GIBBERELLIN INSENSITIVE DWARF1 encodes a soluble receptor for gibberellin. Nature 437, 693–698
Dormancy & Germination
Introduction
After completing embryogenesis and seed maturation, most seeds enter into a state of dormancy, which can be thought of as something like “suspended animation”. Dormancy is characterized by extremely slow metabolic activity brought about by mechanical and/or chemical signals. At this stage, the environment may be sealed out (in the case of coat-imposed dormancy), or the embryo may be under the influence of chemical messages that maintain dormancy. Chief among these chemical messengers is the plant hormone abscisic acid (ABA), a small organic molecule classified as an sesquiterpene and synthesized in plastids.
ABA in Dormancy
ABA plays a number of diverse roles in plants, and its name reflects its discovery based on its involvement in fruit abscission (detachment from the parent plant). ABA also plays a critical role in plant drought response, in part by regulating stomatal pore size. In seeds, ABA concentrations increase with embryo maturation due to its synthesis by the embryo, and results in several responses that promote dormancy. ABA promotes expression of seed storage proteins associated with seed maturity, known as Late Embryogenesis Abundant (LEA) proteins. ABA also promotes the synthesis of proteins associated with desiccation (drying) tolerance, known as heat-shock proteins (HSPs). In addition, ABA also prevents the premature germination of the embryo, known as vivipary.
Dormancy Release
The release of embryo dormancy is associated both with a decrease in ABA synthesis and concentration and an increase in the concentration of a second plant hormone, gibberellic acid (GA). As levels of ABA decrease, the inhibitory effects of this hormone begin to decline. At the same time, this decline promotes the synthesis of GA by the embryo. The increase in GA concentration actively promotes germination, with perhaps its most prominent influence occurring on the expression of several hydrolytic enzymes, including α- and β-amylase. α-amylase is known as an endoglucanase, meaning it hydrolyzes starch chains in mid-chain, while β-amylase is an exoglucanase, acting on the ends of the starch chain to release maltose units (a disaccharide of glucose). As starches are digested by these hydrolytic enzymes, the resulting sugars can be taken up by the embryo and used to fuel growth.
Transition
If high levels of ABA promote dormancy, and high levels of GA promote germination, how does the embryo “know” when to make the switch? There are a number of factors, both intrinsic and external, that influence this decision. External cues such as water availability and chilling exposure have been shown to influence germination, and internal cues related to seed maturation, known as afterripening, are also important in some species. Because plants are photosynthetic autotrophs, waiting to germinate until the light conditions were optimal would seem like an adaptive response, and in fact many plants use light as a cue to regulate germination. In particular, plants use the phytochrome photoreceptor to determine the quality of light available in the environment. Locations that are rich in high-energy light promote germination through the phytochrome signaling pathway, while locations predominated by shade inhibit germination.
Seed Structure
Introduction
Seeds are amazing structures, capable of protecting an embryonic plant for hundreds of years in a state of suspended animation, awaiting the onset of favorable conditions to initiate germination. The seed represents a pause in the lifecycle of plants that is so effective, it conferred a major selective advantage on those species having it, the earliest of which arose in the late Devonian (417-354 Mya) and came to dominate by the early Mesozoic.
Seeds possess a number of improvements over spores that are adaptive, including a seed coat, multicellular embryonic seedling, and a significant reserve of energy. The thickness and material making up the seed coat varies from species to species and plays a role in the particular kind of dormancy experienced by a seed. The nature of the coat also influences the kinds of signals that are able to reach the embryo and release dormancy.
Seed Coat
Plants producing thick, lignified seed coats typically undergo coat-imposed dormancy, characterized by the physical exclusion of light and water, extremely limited gas exchange between the embryo and the environment, and the maintenance of high concentrations of the dormancy-promoting hormone abscisic acid. Some seeds having this kind of dormancy can endure for hundreds of years and remain viable [1]. These seeds are “hermetically sealed” packages, and must be broken open in order for germination to proceed. This breakdown may occur through the activity of animals (including exposure to digestive acids), exposure to freeze-thaw cycles, or other weathering processes.
Plants not having a thickened coat remain capable of dormancy, but rather than the physical restriction of exchange, dormancy arises from production of and response to particular chemical messages. In actual fact, both kinds of dormancy use chemical signaling to promote or release dormancy, but seeds with thickened coats have an additional mechanism for protecting the dormant state of the embryo.
Energy Supply
In addition to a seed coat, all seeds contain a supply of energy contributed by the parent. Depending on the kind of plant, this energy supply may take one of two different forms. In some plants the energy deposited by the parent remains in a large, starchy mass known as endosperm. Grasses (Family Poaceae) such as wheat (Triticum aestivum), maize (Zea mays), and rice (Oryza sativa) all maintain a prominent endosperm throughout seed maturation and dormancy. It is the endosperm, in fact, that we are eating when we eat grains like rice and wheat. In other plants, the endosperm may be absorbed fully by the developing embryo and stored in specialized seed leaves called cotyledons. Plants such as beans (Family Fabaceae) are representative of this strategy.
Embryo
The embryo is the result of fertilization, or syngamy (fusion of gametes). Upon the fusion of the sperm nucleus with the egg nucleus inside the ovule, a single-celled zygote forms and begins developing into the embryo. As early as the first division of the zygote, polarity has already been established that will govern the apical-basal axis of the embryo. The embryo is fully dependent upon the parent plant for nutrition during seed maturation and early seedling establishment, until the seedling becomes photosynthetic and therefore autotrophic (self-feeding). Before this, the embryonic plant is in fact heterotrophic, benefiting from the investment of photosynthate from its parent.