Introduction
As previously discussed, high levels of ABA promote embryo dormancy, while a reduction in ABA concentration leads to an increase in GA biosynthesis and the promotion of germination. But how does the concentration of small molecules make a difference in the cell? These molecules have specific cellular receptors, capable of tightly binding the hormone and activating a signal transduction cascade. The end results of the cascade differ depending on the hormone, but almost always lead to a change in gene expression.
ABA perception
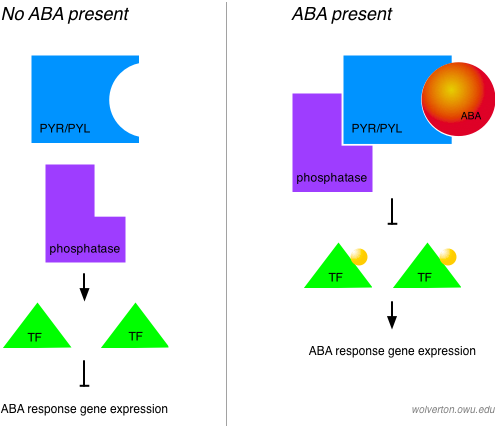
At least one ABA receptor has been identified to date in plants. The PYR1 gene encodes a protein that can bind ABA and inactivate a protein phosphatase in the PP2C class. Protein phosphatases remove phosphate groups from target proteins, often inactivating them. The result of inactivating this phosphatase is to stimulate ABA responses, the earliest of which include activation of ABA-specific transcription factors. So we can think of ABA as inhibiting an inhibitor of ABA signaling — when the inhibitor (a PP2C protein) is inhibited, ABA signaling can proceed.
A receptor for ABA has been sought for many years, but had eluded researchers until recently. The PYR1 gene was isolated in a screen for mutants that showed resistance to a synthetic compound called pyrabactin, an agonist of ABA responses. This kind of approach, called chemical genetics, is one way to side-step the functional redundancy shown by many plant genes due to genome duplication, and is described in more detail by the Cutler Lab, which successfully employed this approach to uncover PYR1.
GA perception
Recent evidence suggests that a protein called GID1 acts as a GA receptor. When GA binds to GID1, it causes the targeted degradation of a group of proteins known as the DELLA family that normally represses GA responses. By getting DELLA proteins out of the way, the cell can begin to mount a response to the presence of GA. This system of targeted degradation uses a pathway known as the 26S proteasome, a large, multi-subunit cellular garbage disposal. To guard against accidental recycling of proteins, the garbage disposal only accepts proteins with a special tag attached. The tag is itself a small protein called ubiquitin, and the closely regulated process of adding the tag to a target protein is called ubiquitination. The GA receptor GID1 is itself a part of the ubiquitinating machinery and acts by causing the ubiquitination of DELLA proteins. To help visualize this process, I have created an animation showing the interaction of these participants in GA signaling.
Responses to these 2 hormones during dormancy and germination are representative of 2 kinds of signal transduction pathways — kinase cascades and ubiquitin-mediated proteolysis. Both kinds of pathways play crucial roles across the domains of life in mediating responses to signals, and there are many more examples of each within the plant kingdom.
References
-
S. Y. Park, P. Fung, N. Nishimura, D. R. Jensen, H. Fujii et al. (2009) Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 324, 1068-1071.
-
Y. Ma, I. Szostkiewicz, A. Korte, D. Moes, Y. Yang et al. (2009) Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 324, 1064-1068.
-
Ueguchi-Tanaka, M. et al. (2005) GIBBERELLIN INSENSITIVE DWARF1 encodes a soluble receptor for gibberellin. Nature 437, 693–698