Light is one form of electromagnetic radiation. As such, it occupies a small slice of the electromagnetic spectrum, which also includes forms of radiation having greater energy than visible light, such as gamma rays and x-rays, and other forms having less energy than light, such as infrared radiation and microwaves. Each of these is characterized by two physical parameters that describe the quality of radiation: wavelength (λ) and frequency (ν). Visible light includes radiation having a wavelength between approximately 400 nm and 700 nm. Because the speed of radiation, c, is a universal constant, wavelength and frequency always vary inversely with each other and their product is equal to c.
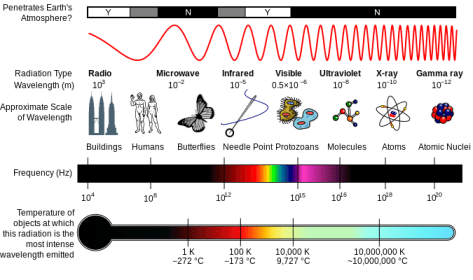
A diagram of the Milton spectrum, showing the type, wavelength (with examples), frequency, the black body emission temperature.
By Inductiveload, NASA [GFDL (http://www.gnu.org/copyleft/fdl.html) or CC-BY-SA-3.0 (http://creativecommons.org/licenses/by-sa/3.0/)%5D, via Wikimedia Commons
Because light is a form of energy, and because we are interested in understanding how light energy is transduced into chemical energy, it is important to understand the relation between light quality and energy. Comparing blue light (450nm < λ < 475nm) with red light (620nm < λ < 730nm) for example, note that blue light has a smaller wavelength and therefore a greater frequency than red light. Even though the speed of light is a constant, the energy is not — energy varies in direct relation to frequency. So in the above example, blue light contains greater energy than red light of a given fluence rate.
In addition to being described in terms of its wave-like properties, the energy in light exists in discrete packets (called photons) rather than as a continuous stream. Photons are measurable down to the individual one, known as a quantum. As we will see next, the fact that light travels in photons, each having a predictable quantum of energy, helps us understand the events surrounding light absorbance by molecules and the transduction of energy into a chemical form.
Light Interacts with Molecules
As you look around the world, you see objects of all different colors. The production of those colors, and in fact our ability to perceive them, both rest on the fact that visible light can interact with matter in specific, predictable ways. Whether a particular frequency of radiation interacts with a particular molecule depends on the structure of the molecule — the arrangement of atoms in space. In the same way an aerial antenna depends on the precise spacing of its elements to intercept radio waves (another form of electromagnetic radiation) of specific frequencies, so too are biological molecules ‘tuned’ to specific frequencies of light to maximize energy transduction. In the case of the photosynthetic pigments known as chlorophylls, their molecular structure allows them to absorb blue and red wavelengths, but not yellow, green, or orange. While most plants have pigments that absorb yellow and orange, green light goes unused by the plant, being either transmitted or reflected.
When a pigment molecule intercepts a photon for which it is receptive, the energy of the photon is conserved by a rearrangement of electrons in the molecule. This rearrangement results in the boosting of an electron from the ground state to an excited state. Sometimes, the excited state is dissipated through the re-emission of light energy, known as fluorescence. Other times, if there are like molecules in close proximity, the excited state can be passed among them through resonant energy transfer. Under certain highly specialized circumstances, the excited state can represent the first step toward a photochemical reaction in which the electron departs the excited state molecule and reduces a nearby electron acceptor. Finally, the excited state may be dissipated through a process called radiationless decay, which releases heat to the surroundings. Each one of these processes plays an important role in photosynthesis.